

Beginning in late July 2026, significant new tariffs are expected on imported patented drugs and key ingredients under Section 232 of the Trade Expansion Act. These tariffs – up to 100% for some manufacturers – are intended to…

Pharmacy Benefit Managers (PBMs) are often discussed in terms of cost – but their value extends far beyond savings. On average, PBMs help patients and employers save over $1,100 per person each year by negotiating drug prices…
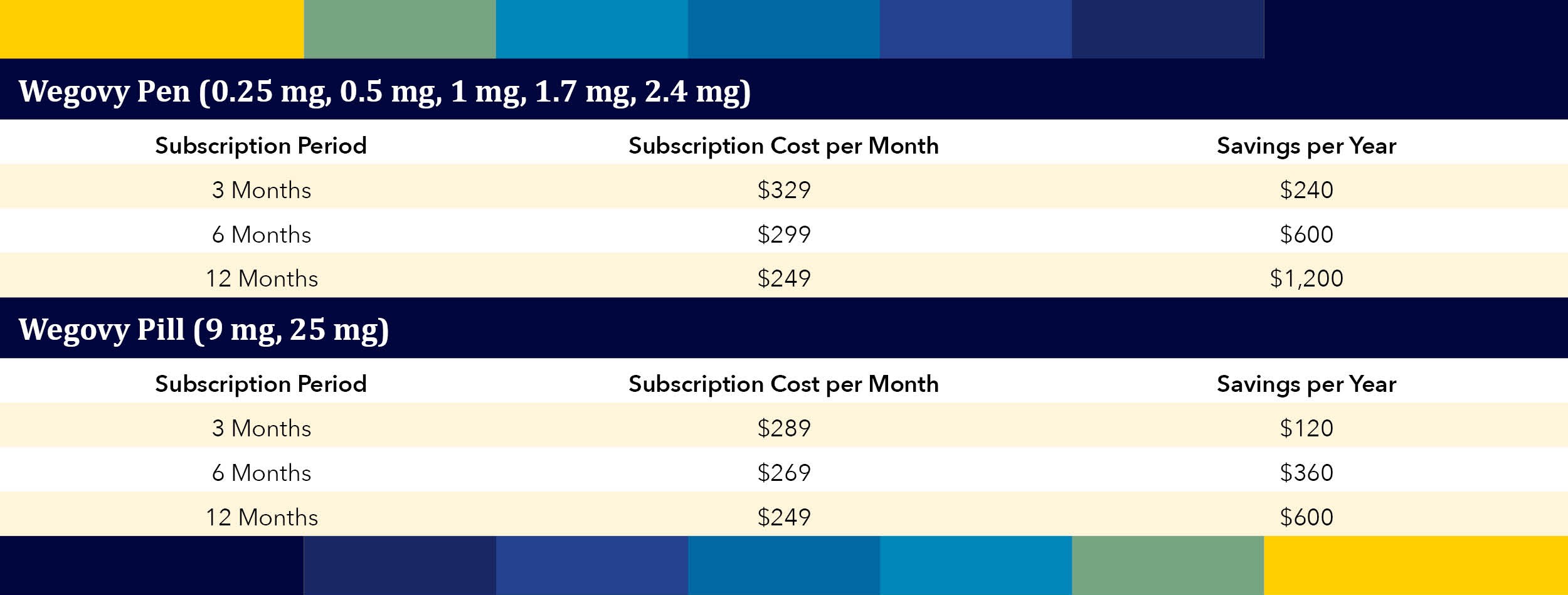
On April 1, 2026, the FDA approved Eli Lilly’s Foundayo, the second oral GLP‑1 for weight loss – offering fewer administration restrictions and achieving one of the fastest new‑drug approvals in decades.…

On March 26, 2026, the FDA approved Awiqli® (insulin icodec abae) from Novo Nordisk – the first once‑weekly basal insulin for adults with type 2 diabetes. This milestone represents a meaningful shift in…
Novo Nordisk recently received FDA approval to bring its oral semaglutide formulation, Rybelsus, under the Ozempic brand – aligning oral and injectable semaglutide for type 2 diabetes under a single, highly recognized name…





